Menu
-

Home
Homepage
KidzSearch
-

Games
Play Fun Mobile Games.
Games
-

KidzTube
Cool Videos for Kids.
Videos
-

KidzTalk
Talk About Anything
Q&A
-

KidzNet
Social Network for Kids
KidzNet
-

Pictures
Explore Popular Images.
Pics
-

Top Sites
Explore the Best Sites for Kids.
Websites
-

Music
Listen to Streaming Music for Kids.
Radio
-

Cool Facts
Facts for Kids.
Facts
-

Kids News
Read About Cool Topics.
News
-

Encyclopedia
Browse Over 200,000 Student Articles.
Wiki
-

Boolify
Learn How to Search with Boolify.
Boolify
-

Mobile Apps
Discover KidzSearch Apps
Apps

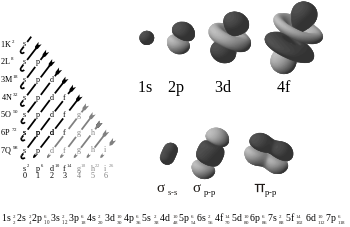
An electron configuration is the arrangement of electrons within an atom. The electron configuration describes where the electrons are inside orbitals. The structure of the Periodic table of elements is partly based on electron configuration.
There are four kinds of electron configurations: s, p, d, and f orbitals. Each orbital can house a maximum of 2 electrons. S orbitals are roughly sphere-shaped, p orbitals are polar and shaped like a dumbbell, d orbitals are usually shaped like a four-leaf clover, and f orbitals form a mathematically complex shape. Orbitals can have different energy levels. For example 1s is a low energy sphere-shaped orbital while 3s is a higher energy sphere-shaped orbital. An atom can have more than one orbital; in fact, all except hydrogen do. Similarly, atoms can have more than one of each kind of orbital.
- Related to electron configuration
- electron configuration periodic table
- electron configuration of gold
- electron configuration gold
- electron configuration definition for kids
- electron configuration table
- electron configuration for argon
- electron configuration for lanthanum
- electron configuration for sulfur
- watch electron configuration videos on kidztube
- play electron configuration games
Post It On KidzTalk Homework Help
Report a search problem
home contact us settings advertise terms/privacyabout usteacher forum
View All
- COMPANY
- contact us
- settings
- advertise
- terms/privacy
- about us
- RESOURCES
- api services
- link to us
- add a site
- media/press
- education daily journal
- blocking websites
- teacher forum
- affiliate program
- make us your default search
- search help
- kidzsearch apps
- voice search
- LINKS
- home
- kidznet
- kidznet directory
- kidztalk
- kidztube
- games
- images
- top sites
- music
- facts
- news
- wiki
- boolify
- SOCIAL
desktop version
Powered by Google SafeSearch
Copyright 2005-2024 KidzSearch.com